Evidence-informed guidelines and studies hold the insights to remove bias and close health equity and outcomes gaps – the work of The Prostate Cancer Foundation advances this goal
ConcertAI was started as a company on the premise that definitive real-world data, advanced AI solutions, and dedicated scientific talent could improve the outcomes of all patients. Yes, too often we see biases, passivity, or a combination of both of bringing the power of data and evidence to bear to both lower the risk of disease, achieve earlier diagnoses, and select the treatments the patient is likely to respond to best. So, it was notable at the recent American Society of Clinical Oncology Genitourinary Cancers Symposium that a clinical panel formed by The Prostate Cancer Foundation provided their read-out on the state of diagnosis and treatment of prostate cancer for Black men with a new set of guidelines that substantially change care across primary, urological and oncological settings.
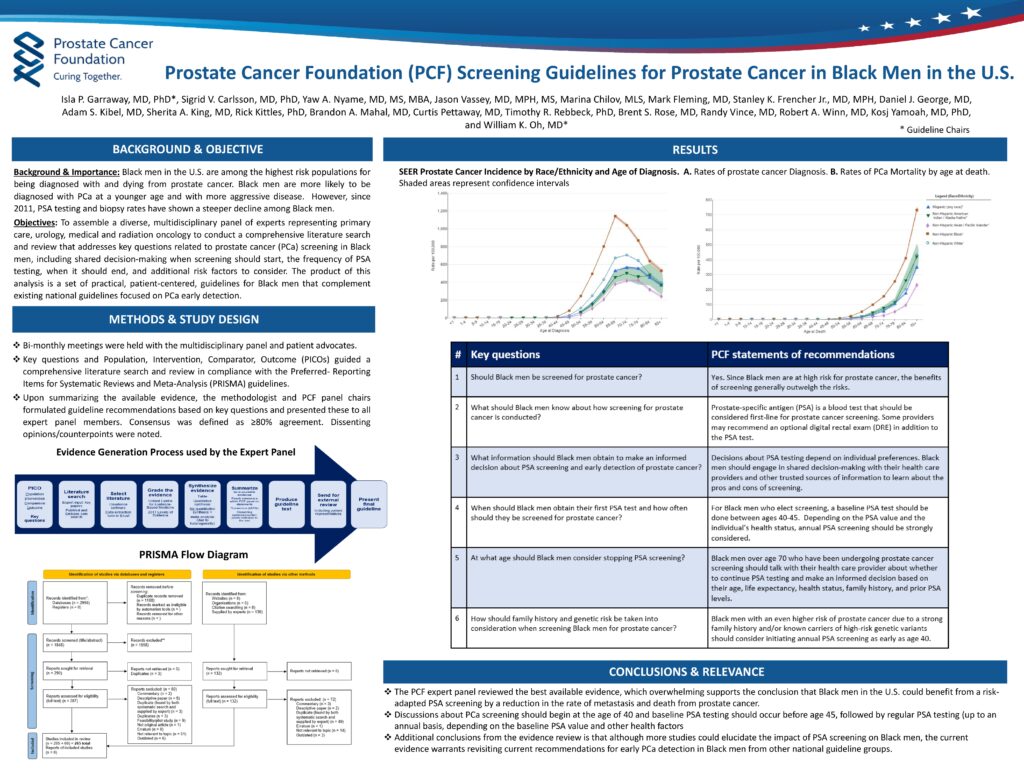
Black men have historically been under screened and diagnosed relative to other racial groups, yet their risks are higher. The US Preventive Services Taskforce current recommendations, being re-evaluated, set PSA screening to start at age 55. The task force informs insurance coverage and is not part of any specific US government agency. The American Cancer Society recommends average-risk men to begin screening at ages 50-55, and lowers this to 45 for those at a higher risks, including Black men and those with immediate relatives diagnosed with prostate cancer occurring before age 65. Together these guidelines trended towards majority population criteria with all other racial, ethnic, and familial groups exceptions and where individual practitioner discretion could prevail. Unfortunately, the lack of formal and definitive guidance has resulted in inconsistent and broadly under-practiced screening for Black men.
Now, based on a comprehensive review of almost 300 published studies, a panel of 18 experts provided an 80% consensus set of new guidelines for the surveillance, diagnosis and treatment of prostate cancer in Black men. The new guidelines recommend a baseline PSA testing beginning at age 40-45, sooner than any current guidelines recommend. The guidelines further note that the baseline should be followed by regular screening intervals, preferably annually, through age 70. It is estimated that lowering the recommended age for baseline prostate-specific antigen (PSA) would reduce prostate cancer deaths by about 30% in Black men without significantly increasing the rate of overdiagnosis and over-treatment. The Foundation made the following specific recommendations:
- Since Black men are at a high risk for prostate cancer, the benefits of screening generally outweigh the risks.
- PSA testing should be considered first line for prostate cancer screening for Black men. Some providers may recommend an optional digital rectal exam in addition to the PSA test.
- Black men should engage in shared decision-making with their healthcare providers and other trusted sources of information to learn about the pros and cons of screening.
- For Black men who elect screening, a baseline PSA test should be done between ages 40 and 45. Depending on the PSA value and the individual’s health status, screening should be strongly considered.
- Black men over age 70 who have been undergoing prostate cancer screening should talk with their healthcare provider about whether to continue PSA testing and make an informed decision based on their age, life expectancy, health status, family history, and prior PSA levels.
- Black men who are at even higher risk due to a strong family history and/or known carriers of high-risk genetic variants should consider initiating annual PSA screening as early as age 40.
ConcertAI has been focused on Prostate Cancer research over the last five years, integrating data from the urological and oncology settings in order to have the fullest view of clinical care, treatment approaches, and outcomes assessments. Our data extends from the earliest stage of disease to late stage and metastatic. In our data, we see the results of not having guidelines aligned to the risk of Black men, and the consequent more advanced diseases stages for initial diagnosis and treatment. We are active with leading biopharma in advancing new treatment approaches, focusing on the diversity of the trial participant population in assurance that data is generated for all impacted populations. In fact, Jennifer Rider, ScD, who leads our RWE regulatory and non-regulatory research, is a former Prostate Cancer Foundation Young Investigator and has been focused on prostate research for almost two decades. It is with this context and appreciation of the current deficiencies of prostate care for Black men, that we applaud the recent work and new guidelines.